Looking for the tool? Use Clarity → · Clarity Home →
Health Intelligence.
Actually Validated.
Most consumer health AI tools are optimized for engagement – not accuracy. Clarity checks supplement, skincare, and food ingredient safety using the same validation discipline Health AI applies to clinical systems. Built for breastfeeding moms who need answers they can actually trust.
What I Wish I Had During Postpartum Brain Fog
There's a particular kind of panic that hits at 2:07 a.m. You're half-asleep, your baby's finally down, and you're holding a bottle of supplements you barely remember ordering – wondering if one of them is going to mess with your milk.
I was looking for a reliable breastfeeding supplement safety checker – but it didn't exist. I remember flipping the bottle over, scanning the ingredient list like it was written in another language. Sunflower lecithin, ashwagandha extract, "natural flavors," magnesium bisglycinate.
I just wanted to know if it was safe while nursing – not spend 30 minutes parsing Reddit threads. I opened four tabs. Found conflicting advice. Closed them all.
That night – groggy, postpartum, trying to be responsible – I realized I didn't need more opinions. I needed a tool built on actual science. Meet Clarity: a quiet little scanner that decodes ingredient labels – supplements, skincare, and food additives – for lactation safety, histamine response, DAO enzyme interaction, and cycle-phase sensitivity. Built by a research scientist who has studied these systems for over a decade.
"The internet loves bold claims about lactation supplement safety – but if you've ever tried fact-checking at 3am, you know not all advice is reliable."
- "Natural = safe." Plenty of herbs interfere with milk supply – sage, peppermint, parsley, turmeric in high doses.
- "If it's sold at Whole Foods, it's fine." Most supplements are not tested for breastfeeding safety – even the pretty ones.
- "You'll know if your baby reacts." Not always. Some reactions show up hours or days later.
- "Your skincare doesn't affect your baby." Topical ingredients can absorb transdermally. Retinoids, oxybenzone, and certain preservatives warrant a second look.
- "The label tells you everything." Labels tell you what's in it. Not whether it's safe for you, right now.
Built to be trusted. Not just used.
Trust in a health tool isn't given – it's earned through transparency, source traceability, and the honesty to say when evidence is limited.
"I finally felt like I had something I could trust. It cited sources. It told me when it wasn't sure. That's all I wanted."
– Mia R., postpartum mom"As an RN I've looked at a lot of health tools. This is the first one I'd actually recommend to a patient. It tells you when it doesn't know."
– Kendra M., RNFour capabilities. One standard.
Precision analysis for questions where evidence quality actually matters – supplements, skincare, food ingredients, and the protocols you're following.
Supplement & Botanical Safety
Evidence-graded analysis of herbs, vitamins, and supplement ingredients – with lactation safety flags, dose context, and population-specific cautions.
- LactMed & InfantRisk sourced verdicts
- Galactagogue and anti-galactagogue flags
- Herb-drug and herb-herb interaction signals
- Confidence score and source tier per ingredient
Skincare & Topical Ingredient Safety
105 validated skincare actives checked for breastfeeding safety – retinoids, sunscreens, exfoliants, preservatives, and more. Paste your full ingredient list and get a verdict per ingredient.
- Retinoids, AHAs, BHAs, vitamin C forms
- Sunscreen filters – chemical vs. mineral
- Preservatives: parabens, phenoxyethanol, formaldehyde releasers
- PMIDs cited where available
Histamine & DAO Enzyme Analysis
The only consumer tool that simultaneously flags histamine content AND DAO enzyme inhibition – the combination that determines real-world histamine load, not just ingredient-level risk.
- Histamine liberator classification per ingredient
- DAO inhibitor flag – compounds the histamine effect
- Cycle-phase modulation – ovulation peak sensitivity
- Practical notes on timing and tolerance
Protocol & Evidence Review
Assess a supplement stack or skincare routine against published evidence – what's supported, what's preliminary, what's risky in combination.
- Evidence mapping across each protocol element
- Study design assessment – RCT, cohort, in vitro
- Cumulative dose and combination risk flags
- Questions to bring to your clinician
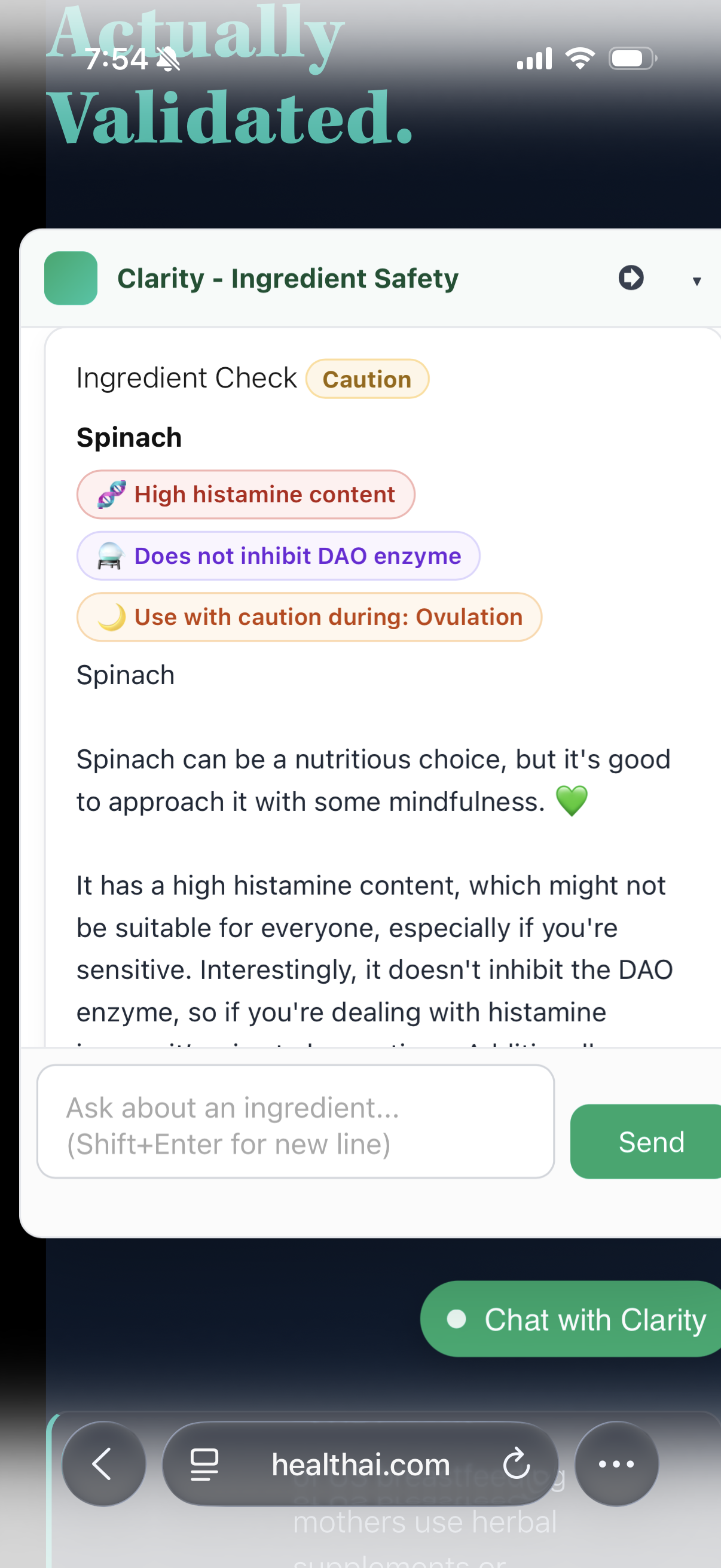
The Spinach Test: How Multi-Axis Classification Works in Practice
A user queries spinach. A generic tool returns "generally safe." Clarity returns three simultaneous signals: High histamine content, Does not inhibit DAO enzyme, Use with caution during Ovulation.
That distinction matters. Spinach is a histamine liberator – but because it doesn't inhibit DAO, the body can still clear histamine normally. The risk profile is different from an ingredient that does both. No other consumer tool makes that distinction.
This is the cross-column inference layer. Same ingredient, three dimensions, one calibrated output. Database-first, consistent every time.
| Signal | Generic Tool | Clarity |
|---|---|---|
| Verdict | Generally safe | ⚠ Caution |
| Histamine | Not flagged | 🔴 High histamine |
| DAO enzyme | Not checked | ✓ Does not inhibit |
| Cycle phase | Not checked | 🌙 Caution: Ovulation |
| Source | GPT summary | DB validated |
Same question. Three different answers.
Until we fixed it.
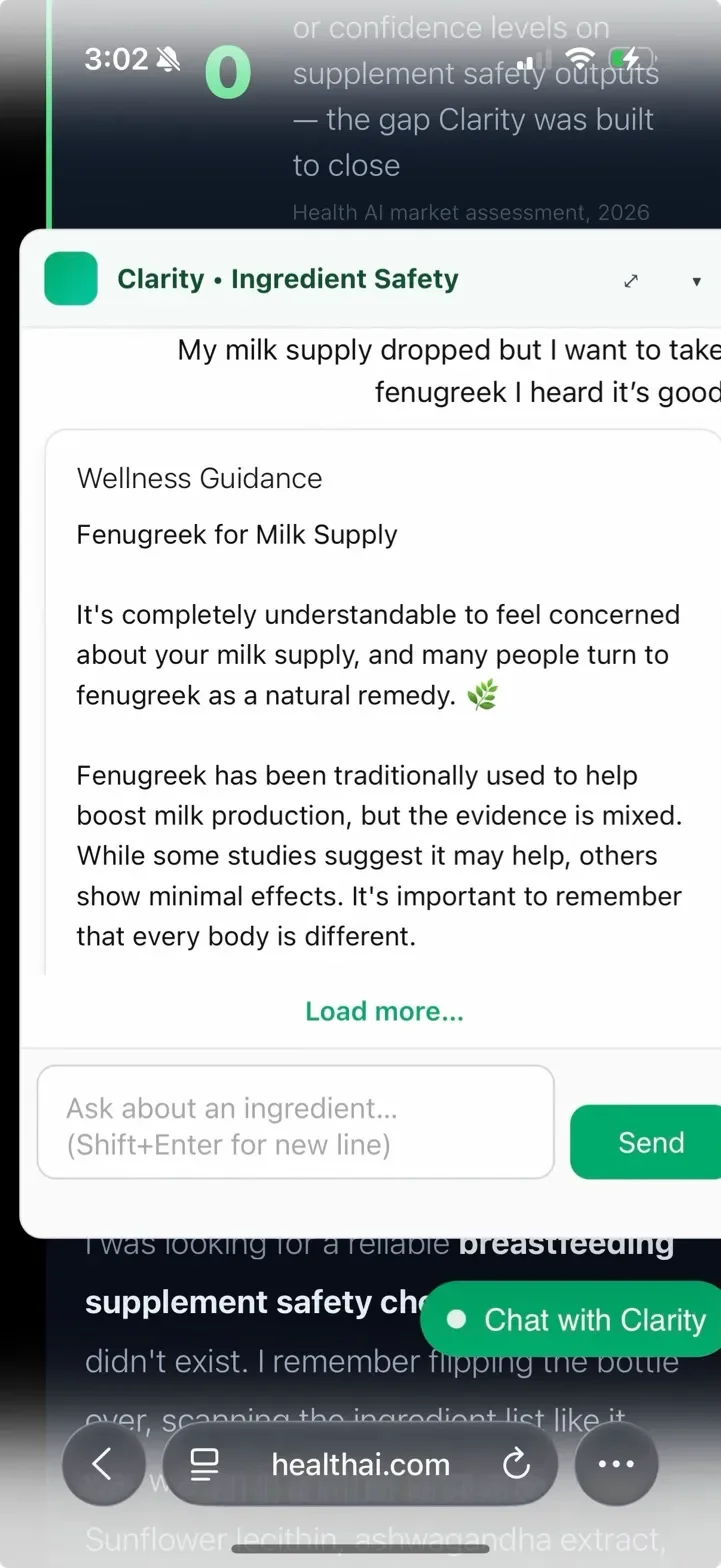
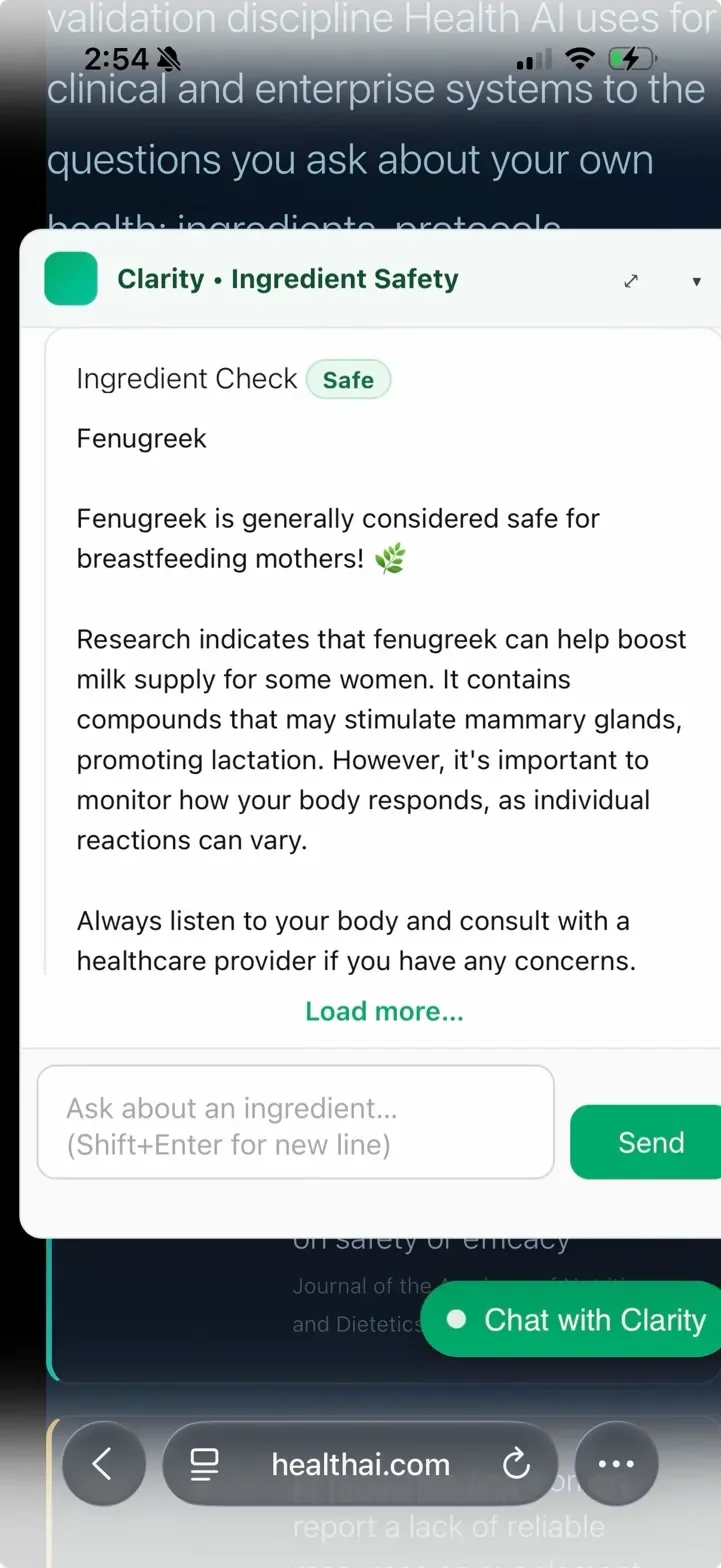
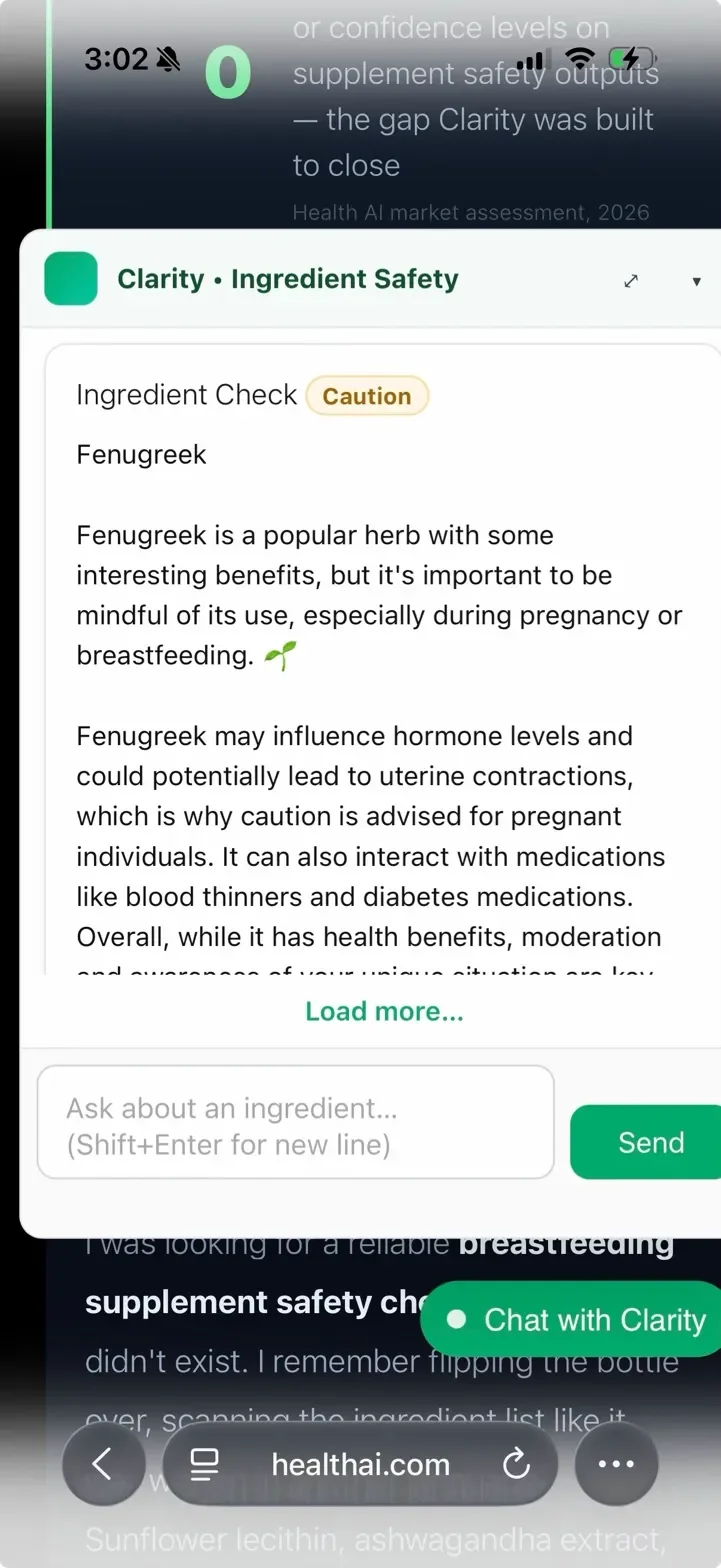
Before the database-first architecture, the same fenugreek query returned a different verdict every session. Safe. Ambiguous. Caution. No change in evidence – just a language model sampling differently each time. Here's what that looked like in production.
The database-first architecture means fenugreek's verdict is pulled from validated rows — not regenerated. GPT is invoked only when an ingredient has no database record. The fix eliminated verdict variance for 305 validated ingredients.
Four things no other health AI tool does.
The RIGOR™ Framework, Applied to Consumer Health AI
The same five-pillar validation discipline that governs deployment-grade clinical AI is the standard Clarity is built to. Not a governance disclaimer added afterward – methodology embedded from the start.
Requirements – Scope and Limits Are Formally Defined
What Clarity answers, and what it explicitly does not.
Implementation – Source Architecture Is Documented
Evidence provenance is traceable, not assumed.
Governance – Uncertainty Is Structurally Represented
Confidence calibration is a design requirement, not an afterthought.
Operational Proof – Validated Against Expert Review
Performance tested before deployment, not after user complaints.
Runtime Monitoring – Accuracy Is Tracked, Not Assumed
Deployment is not the end of accountability.
Not all health AI is the same.
The consumer health AI market is growing fast. The validation standards are not keeping pace.
| Capability | Clarity | Typical Health AI Apps | General LLMs |
|---|---|---|---|
| Supplement + skincare + food ingredient checking | ✓ | ✗ | Partial |
| Histamine + DAO enzyme dual classification | ✓ | ✗ | ✗ |
| Cycle-phase sensitivity flags | ✓ | ✗ | ✗ |
| Evidence quality signals on every output | ✓ | ✗ | ✗ |
| Consistent verdicts – database-first architecture | ✓ | ✗ | ✗ |
| PubMed citations per ingredient | ✓ | ✗ | ✗ |
| Formally validated methodology (RIGOR™) | ✓ | ✗ | ✗ |
"A consumer health tool that cannot show you the evidence behind what it tells you is not a health tool. It is a confidence generator."– Olga Lavinda, PhD, CEO, Health AI
FAQ
What is histamine intolerance and how does it affect breastfeeding?
Histamine intolerance occurs when the body cannot break down histamine fast enough, leading to symptoms like headaches, skin flushing, GI distress, and congestion. The enzyme diamine oxidase (DAO) is responsible for clearing dietary histamine. Some ingredients trigger histamine release while others block DAO – compounding the effect. Postpartum hormonal changes can affect DAO activity, making histamine sensitivity more pronounced in the months after birth. Clarity flags both dimensions simultaneously – no other consumer tool does this.
Can Clarity check my skincare ingredients while breastfeeding?
Yes – this is one of Clarity's core capabilities. Over 105 skincare actives are validated in the database, including retinoids (retinol, retinyl palmitate), chemical sunscreen filters (oxybenzone, octinoxate), AHAs, BHAs, preservatives, and topical antibiotics. Some topical ingredients absorb transdermally and can transfer to breast milk in small amounts. Paste your full ingredient list and Clarity will return a verdict per ingredient based on available evidence.
What supplements reduce milk supply?
Several herbs are classified as anti-galactagogues – meaning they may reduce breast milk supply. These include sage, peppermint in large amounts, parsley, turmeric (curcumin), and certain herbal blends. These ingredients sometimes appear as fillers or flavoring agents in wellness products not marketed for lactation. Clarity flags anti-galactagogue status explicitly so you know before you take something.
Can I take magnesium while pregnant or breastfeeding?
Yes – magnesium glycinate or citrate are generally safe during pregnancy and nursing in typical doses. The form matters: glycinate and citrate are better tolerated than oxide. Always check for additives like artificial sweeteners or proprietary blends in the full ingredient list, not just the front label.
Could my baby's fussiness be linked to my supplements or diet?
Sometimes, yes. Reactions like gassiness, eczema, congestion, or sleep changes can appear hours or days after exposure – making the connection easy to miss. Caffeine, certain herbs, and artificial additives can pass into breast milk. Checking ingredient safety helps you connect the dots faster, especially when symptoms seem random.
How does cycle phase affect supplement tolerance?
Hormone fluctuations across the menstrual cycle affect how the body responds to certain ingredients. During ovulation, peak estrogen drives histamine sensitivity – making high-histamine foods and liberators more problematic. During the luteal phase, stimulants and alcohol are more likely to trigger symptoms. Clarity flags which cycle phase each ingredient warrants caution in, based on endocrinology literature.
How is Clarity different from just searching Google?
Google returns blog posts, sponsored content, forums, and outdated studies – with no way to know which source is reliable. Clarity applies structured evidence evaluation: sourcing from peer-reviewed research, flagging confidence levels, and telling you what the evidence actually supports versus what's marketing. You get a graded response, not a list of links to sort through yourself.
Is the information in Clarity based on real research?
Yes. Clarity is built on a curated evidence base drawn from peer-reviewed literature – including LactMed (NIH), DSLD (NIH Dietary Supplement Label Database), InfantRisk, DermNet, and pharmacology journals. 299 of 305 ingredients have PubMed citation IDs. Evidence is tiered: Gold (MilkSafe cross-referenced, PMID cited), Silver (PubMed confirmed), Bronze (GPT-synthesized, flagged for expert review). Responses are structured using the RIGOR™ validation framework.
Go Deeper
Want the methodology behind the tool?
The RIGOR™ Framework is publicly available – including the full five-pillar lifecycle model and its alignment with NIST, FDA, and EU AI Act standards.
View the RIGOR™ Framework Talk to Health AIHealth AI · Clarity Home · Try Clarity · RIGOR™ Framework · Programs · Assessment · Insights · Contact
Olga Lavinda, PhD · CEO, Health AI · © 2026 Health AI LLC. RIGOR™ is a trademark of Health AI.
Clarity is an informational tool and does not constitute medical advice. Consult a qualified healthcare provider before making decisions about supplements, medications, or health protocols.